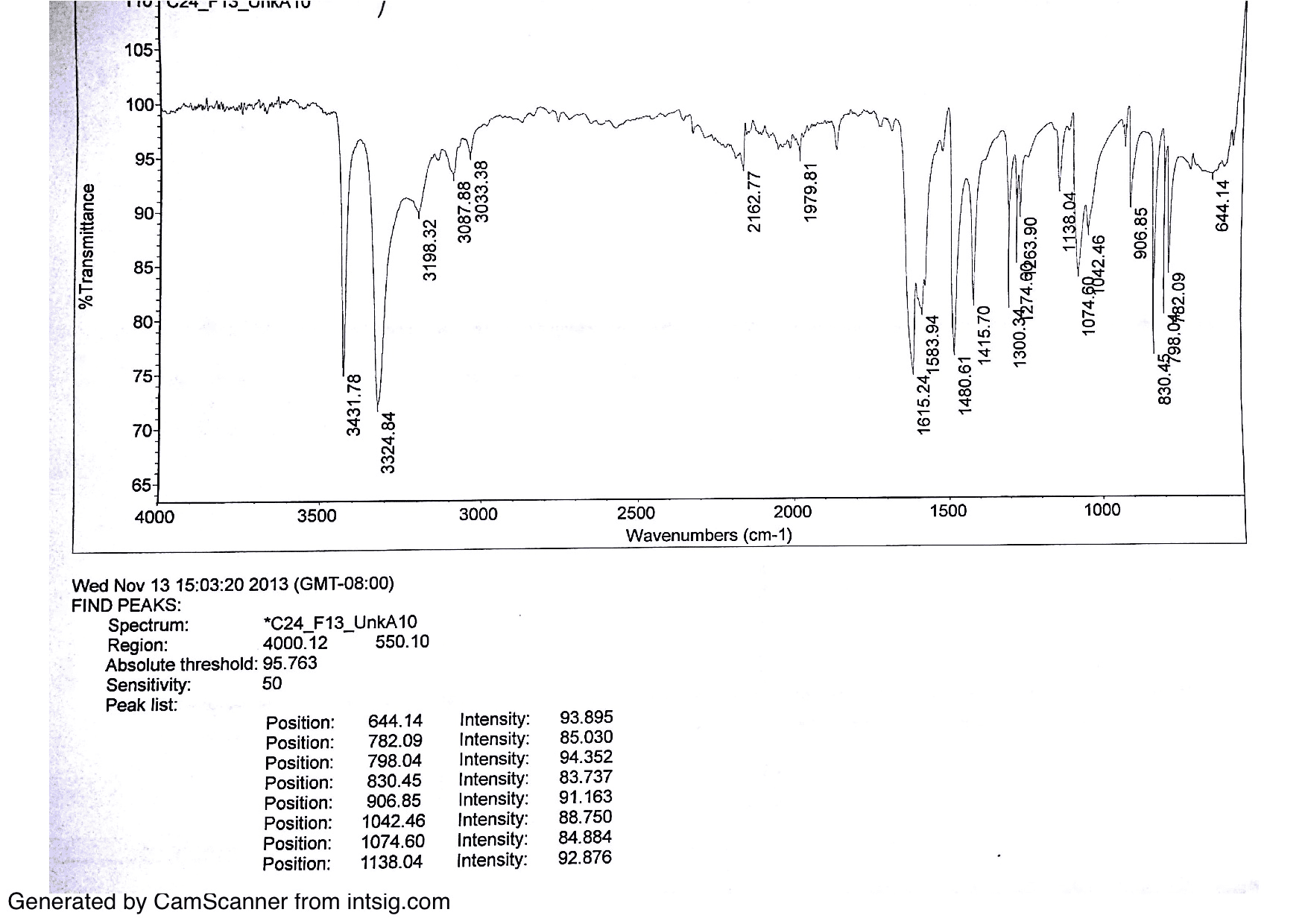
In the infrared region, the commonly used unit is the wavenumber ( v¯), which is expressed in cm- 1, because it is directly proportional to energy ( E) and frequency (v) of radiation, according to the equation: The position of absorption bands in the spectra are presented as wavelength (λ), either as meters or sub-multiples of a meter. There are two types of molecular vibrations: one that changes the bond length ( stretching) and other that changes the bond angle ( bending). The vibration frequencies of a chemical bond depend on the stiffness of this bond (characterized by a proportionality constant termed the force constant) and the masses of the atoms at each end of the bond. For a material to absorb radiation in the infrared region, two conditions should be fulfilled: (1) there must be coincidence (resonance) among the frequencies of the infrared radiation and molecular vibration (2) the natural vibration must cause change in the dipole moment during vibration. Not all possible vibrations within a molecule will result in an absorption band in the infrared (IR) region. It is based on the interaction between electromagnetic radiation and natural vibrations of the chemical bonds among atoms that compose the matter. Therefore, this paper presents the foundations and experimental details that can aid researchers interested in using this technique with ultimate outcomes.įTIR spectroscopy is a widely used technique for investigating materials in the gaseous, liquid or solid phase. The use of FTIR spectroscopy requires certain precautions during sample preparation and method performance in order to obtain appropriate spectral data that will provide an accurate determination of the conversion degree. Among these, FTIR is the most frequently used technique. Techniques such as Fourier transform infrared (FTIR) spectroscopy 3, 13, 14, RAMAN 4, 12, EPR 15 (electron paramagnetic resonance), NMR 9 (nuclear magnetic resonance), DSC 1 (differential scanning calorimetry) and DTA 6, 8 (differential thermal analysis) have been used to determine the DC. In general, the photoactivated dental composites reach a DC ranging from 43 to 75%, basically depending on the composite composition, irradiation intensity and exposure time 5, 11. The limited conversion found in many network polymers is due to restricted mobility of radical chain ends, pendant methacrylate and monomer imposed at high crosslink density 5. The cured composites based on monomers with high molecular weight, as the Bis-GMA or UEDMA, exhibit a considerable number of remaining double bonds 3, 13. Typical composites used in dental restorative applications are composed of at least two dimethacrylate monomers. The degree of conversion (DC) is determined by the proportion of the remaining concentration of the aliphatic C=C double bonds in a cured sample relative to the total number of C=C bonds in the uncured material. The physical and mechanical properties of photo-cured composites are directly influenced by the level of conversion attained during polymerization.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
